Community midwifery model of postpartum care as a path toward equity and better health
April 12, 2024
New research from Ariana Thompson-Lastad and colleagues uses interviews with midwives who provide care in birth centers and home settings in California and Oregon to describe six key elements of the community midwifery model of postpartum care.
New resource: Guide to accurate and compassionate communications about drug and alcohol use during pregnancy
April 09, 2024
Our new resource offers guidance that promotes health and well-being, is grounded in evidence, and tells stories of pregnant people who use drugs and alcohol with compassion, humility and accuracy.
Telehealth medication abortion is just as safe and effective as in-person care
March 11, 2024
As we await a decision on FDA v. Alliance for Hippocratic Medicine from the Supreme Court that could potentially strip access to medication abortion through telehealth services and virtual appointments, new ANSIRH research from The California Home Abortion by Telehealth (CHAT) Study published in ...
A framework for contraceptive agency
February 16, 2024
In a new commentary, researchers with Innovations for Choice and Autonomy identify areas for innovation in defining and measuring contraceptive agency and offer a new framework.
The importance of "booster" sex education
February 14, 2024
Researchers partnered with a Youth Advisory Council to study how a new “booster” sex education called READY, Set, Go! can reinforce and expand upon students’ existing sexual health knowledge.
Black pregnant women's anxiety increased during COVID-19
February 01, 2024
New research found that Black pregnant women’s anxiety increased due to the COVID-19 pandemic, caused by several factors beyond fear of infection.
Self-injected birth control more available during COVID-19, but overall availability remains low
January 17, 2024
New research from Beyond the Pill in Contraception shows that although birth control injections that people can administer themselves increased during the COVID-19 pandemic, the method availability remains persistently low.
Creating an environment where providers can offer the best care
January 04, 2024
Patience Afulani and team tested an intervention to address provider stress and bias in Migori County, Kenya.
Patients with disabilities experience more barriers to reproductive care
January 03, 2024
Using surveys conducted during the COVID-19 pandemic, ANSIRH researchers are studying the prevalence of ableism in reproductive health care and the barriers experienced by people with disabilities.
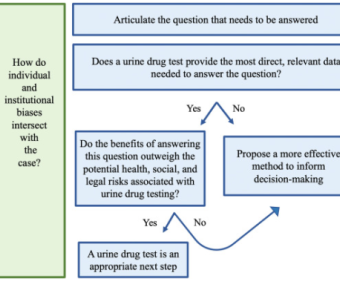
Reconsidering urine drug testing in reproductive health
December 22, 2023
Researchers have created a new framework to assess whether to use urine drug tests in reproductive health settings.